|
For the purposes of the VSEPR model, a double or triple bond is no different in terms of repulsion than a single bond. Molecules can be systematically classified according to the number of bonding pairs of electrons as well as the number of nonbonding, or lone pairs, around the central atom. The model states that electron pairs will repel each other such that the shape of the molecule will adjust, so that the valence electron-pairs stay as far apart from one another as possible. The acronym VSEPR stands for the valence-shell electron pair repulsion model. The valence shell holds the electrons that are involved in bonding and are the electrons shown in a Lewis structure. Depending on how many of the clouds are lone pairs, the molecular geometry will be tetrahedral (no lone pairs), trigonal. To minimize repulsions, four electron clouds will always adopt a tetrahedral electron geometry. To remove all geometrical measurements from the display right-click within the 3D viewer and select Measure, followed by either Clear distances, Clear angles or. The VSEPR model can be used to predict the shapes of many molecules and polyatomic ions, but it gives no information about bond lengths and the presence of multiple bonds. 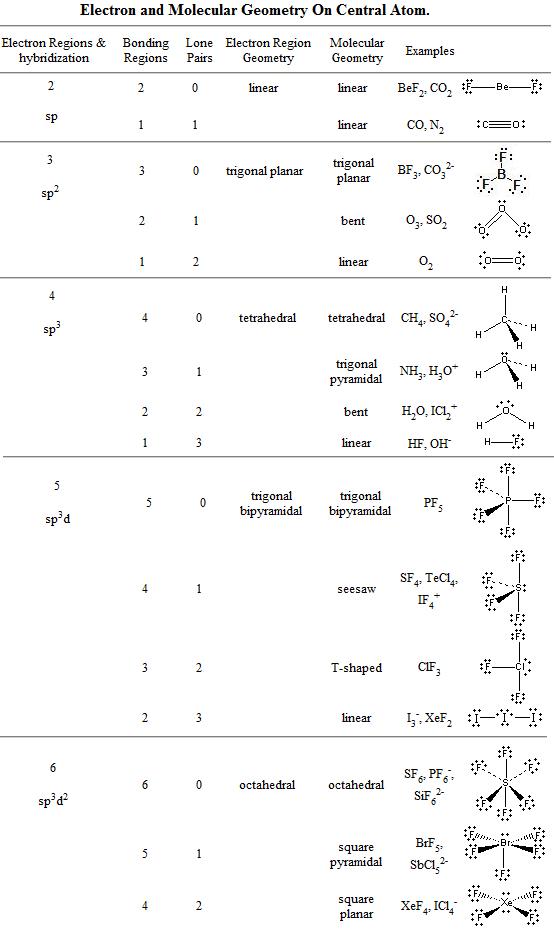
The valence shell is the outermost electron-occupied shell of an atom. In this video, we apply VSEPR theory to molecules and ions with four groups or clouds of electrons around the central atom. Dipoles and Electrostatic Surfaces XeF4, ClF3 and CCl3Br VSEPR CH4 Methane VSEPR ClF3 Chlorine Trifluoride VSEPR H2O Water VSEPR NH3 Ammonia VSEPR PF5. In this video, we apply VSEPR theory to molecules and ions with five groups or clouds of electrons around the central atom. A covalent bond is formed when non-metal atoms share their valence electrons, which they do in order to achieve filled valence orbitals like their nearest. They developed a new approach based on earlier work by other scientists that incorporated a consideration of electron pairs in predicting three-dimensional structure. To practice predicting molecular shapes (using VSEPR theory) and molecular polarity Non-metal atoms bond covalently, resulting in the formation of either neutral molecules or polyatomic ions. Using the VSEPR theory, the electron bond pairs and lone pairs on the center atom will help us predict the shape of a. An electron group can be an electron pair, a lone pair, a single unpaired electron, a double bond or a triple bond on the center atom.  
The problem was that this theory gave an incorrect prediction of bond angles for many compounds. VSEPR focuses not only on electron pairs, but it also focus on electron groups as a whole. The theory at that time relied on hybrid orbitals to explain all aspects of bonding. Nyholm recognized that the current model for explaining bond angles did not work well.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
